Using paper and carton boards from responsibly managed forests, developing simpler packaging solutions from mono-materials and consistently implementing recyclability for folding cartons, labels and package leaflets – these are some of the approaches that make it possible to meet these challenges.
At Faller Packaging, we have incorporated a sustainable approach into our work for many years now and have identified five important characteristics of secondary packaging materials that we believe are crucial for the packaging of sterile products:
- Compliant
- Sustainable
- Customised
- Flexible
- Efficient
Sustainable packaging solutions for the pharmaceutical industry must comply with nationally and internationally applicable regulations in order to securely protect medicines against counterfeiting. In the EU, pharmaceutical manufacturers have been required to provide their products with corresponding security features since 2019. This also applies for other important markets such as the United States, China and Russia. Manufacturers are required to serialise their folding cartons or labels. In addition, in order for packaging to be legally compliant, it must also include a tamper-evident seal that visually protects against tampering, a package leaflet with all the important information and labels for sensitive sterile products. Additional brand protection can be provided by other overt or covert security features.
In the case of primary packaging for parenterals – vials, syringes and cartridges – special designs are usually used instead of standard folding cartons. The special packaging with inserts can be adapted exactly to the respective product or set to be packaged. With modular packaging concepts, different injectable medicines can be packaged with only one pack size and the respective matching insert. This is advantageous for pharmacists, who can thus quickly adapt their production processes to the life cycles of their products. There are myriad possibilities, as the carton board inserts offer the best possible protection and break resistance. These concepts also facilitate flexible and secure packaging processes using conventional cartoning machines.
Sustainability is currently the single most important factor influencing developments in the packaging sector. Sustainable management has been a cornerstone of our corporate culture for many years. That's why we only use fibre-based, fully recyclable mono packaging for our products: For folding cartons, subdivisions and inserts, we use 100% certified and sustainable raw materials. Labels are laminate-free (where it is feasible to do so) and are fully recyclable. Specially developed lightweight papers are used for package leaflets. This allows us to achieve the same print image with up to 20% less ink than conventional pharmaceutical papers. Only paints and varnishes without heavy metal compounds, mineral oils and aromatic solvents are used.
In addition to all the functional and safety-relevant properties of pharmaceutical packaging, the issue of efficiency is also at the forefront of procurement. More and more specific forms of treatment are creating an increased demand for very small lot sizes. At the same time, manufacturers are bringing their preparations to market faster than ever before due to increasingly short product life cycles. Secondary packaging can be procured more efficiently by reducing the administrative effort involved in purchasing. As a true one-stop shop, we manufacture folding cartons, labels, combination products and package leaflets in our own production facilities and assemble them into integrated products as required. We are continually working on our energy and raw material consumption as well.
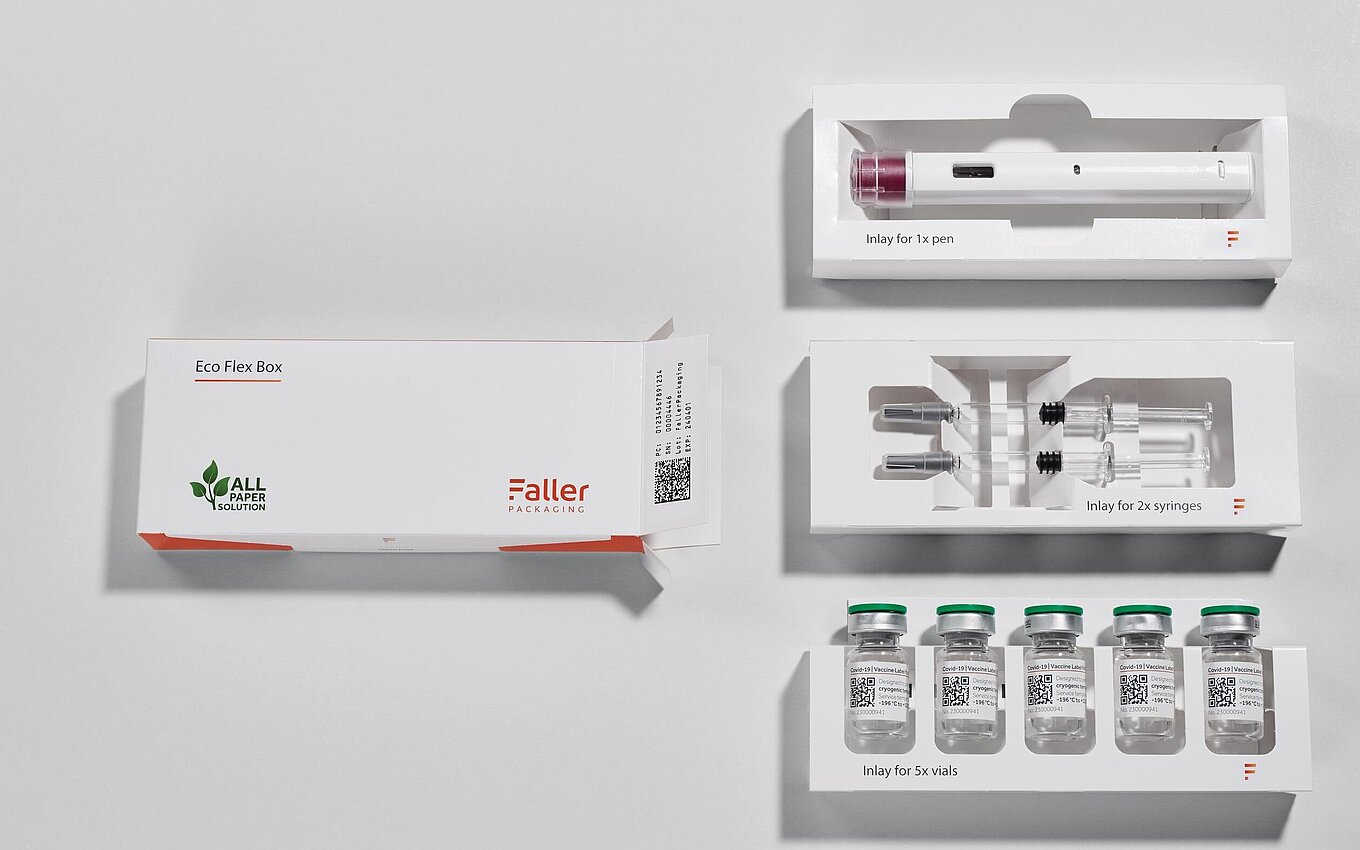
Sophisticated and sensitive sterile products must be safe to transport and store. In order to meet these demands, secondary packaging, i.e. the outer packaging of vials, ampoules or syringes, plays an important role. Customer-specific packaging solutions with custom-fit inlays protect them from blows or falls as well as against pressure. Instead of the usual inserts made of plastic, we work with resource-saving and renewable mono-materials for these forms of environmentally friendly pharmapackaging. The inserts made of recyclable carton board also prevent glass-to-glass contact of the components and ensure sufficient product, breakage and transport protection. We are happy to adapt this special design to the specific sterile product to be packaged according to customer requirements.
Eco Flex Box: environmentally friendly - flexible - modular
Discover all our product features with interactive media
A harmonised concept of machine and packaging
A harmonised concept of machine and packaging
In addition to efficient procurement, the way different forms of packaging interact with the packaging machines is also crucial: Material and machines must fit together optimally so that users can also process different products or combination packaging safely and efficiently on one line. This is why we work closely with the packaging technology specialist ROTZINGER PharmaPack

Conventional packaging concepts usually include inserts made of plastic. Our alternative is an ecologically friendly type of packaging that uses inserts made of carton board.
Faller Packaging works with resource-saving and renewable mono-materials for these forms of environmentally friendly pharmapackaging. The inserts made of recyclable carton board offer optimal product affixing.
It goes without saying that easy mechanical product feeding and a hassle-free packaging process are guaranteed.
Faller Packaging as a partner for sustainable packaging concepts
Sustainable business management is increasingly coming into focus and is shifting from a fringe issue to an indispensable ‘must-have’. For this reason, holistic and forward-looking packaging concepts will be increasingly in demand in future, and the need for flexible and sustainable pharmapackaging will only continue to grow. As a one-stop shop and with our broad, international positioning along the entire supply chain, Faller Packaging is ideally equipped to meet these challenges for folding cartons, package leaflets and labels.
![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/3/2/csm__DSC0924_d304bf6a09.jpg)